|
Now that your DNA fragment has been cloned into a donor plasmid, it can be rapidly shuttled into any compatible Gateway® Destination vector, which contain attR sites via LR clonase enzymes. The entry clone now has recombined attL sites flanking your DNA fragment of interest. 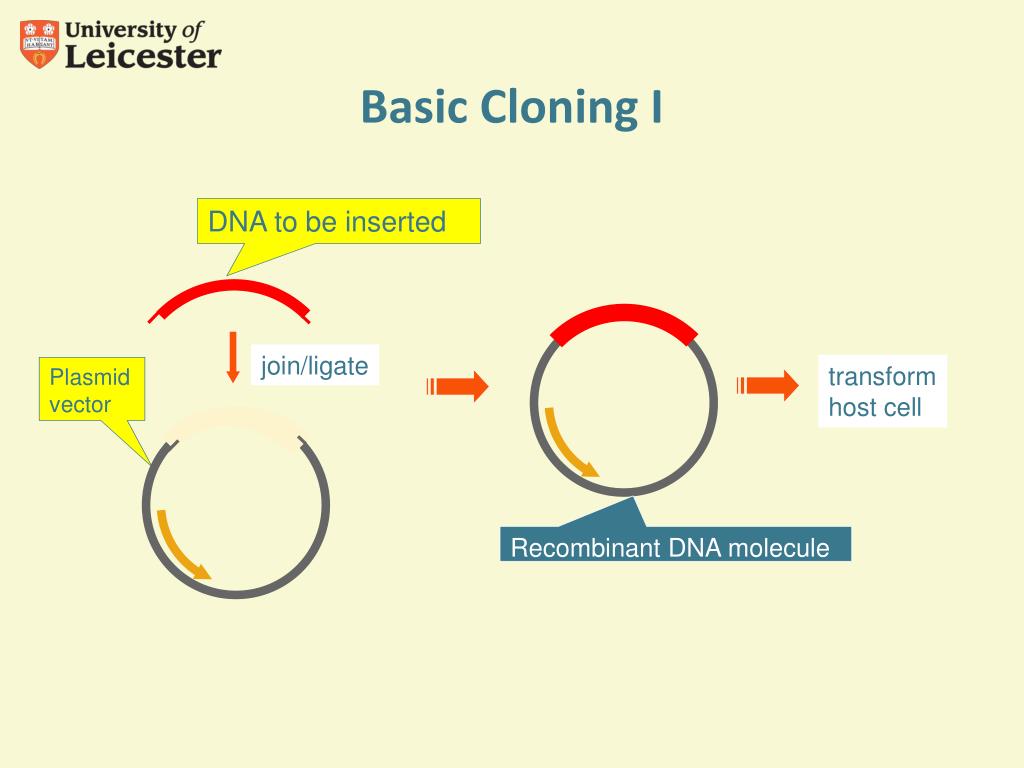
This fragment can then be cloned into a gateway donor plasmid which contains compatible attP sites via a proprietary BP clonase (creating an entry clone). To do this your DNA fragment must first be amplified with specific Gateway attB1 and attB2 sites attached to the 5’ and 3’ ends of DNA sequence. To utilize this approach, the fragment of DNA that you would like to clone into a plasmid must already be surrounded by specific recombination sites (in this regard, not so dissimilar from restriction enzyme cloning). The benefit of Gateway® is that moving a piece of DNA from one plasmid into another is done via a single recombination reaction, drastically simplifying the process and reducing the amount of time required for cloning. Gateway® cloning is a recombination based cloning method. Gateway® Recombination Cloning Gateway cloning (Image from Plasmid 101: Gateway Cloning) (Image from Plasmid 101: Restriction cloning) Restriction cloning of your gene of interest (YGOI) into a recipient plasmid. You can find a protocol for restriction cloning and an in-depth breakdown of restriction digests on our website. Restriction enzyme cloning also leaves behind a short scar in the DNA sequence and can be time consuming compared to other cloning methods. It is important to note that restriction enzyme target sites can be repeated throughout a specific DNA sequence, which can make it difficult at times to identify compatible restriction enzymes, that cut your insert or backbone at only the desired location for your cloning project. Due to their short length, it is also easy to add restriction sites to any piece of DNA during PCR amplification, allowing for it to then be digested and ligated into your desired plasmid. You can easily move (subclone) any piece of DNA that already has restriction sites on either side of it into any plasmid that has the same sites in the same orientation within its multiple cloning site. 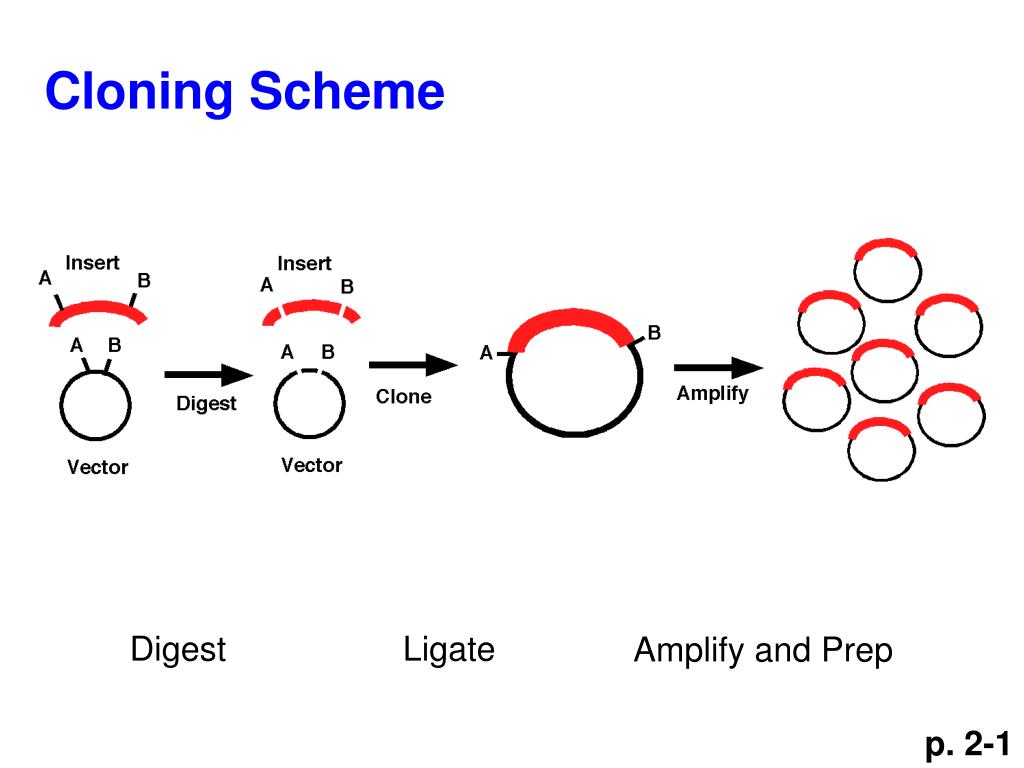
Given its prevalence, the vast majority of plasmids used for DNA cloning and expression contain several popular restriction enzyme sites. They also cut specific target sequences, that range from 4 to 13 base pairs, and produce predictable resulting ends in the DNA fragments. Restriction enzyme cloning benefits from the hundreds of available enzymes, many of which are relatively cheap. Two pieces of DNA that have complementary overhangs, or which are both blunt-ended, can then be fused together during a ligation reaction with T4 DNA ligase. In restriction cloning, scientists utilize specific restriction enzymes to cut dsDNA of interest into fragments containing precise 5' or 3' single-strand overhangs (sticky ends), or no overhang (blunt ends). Restriction enzymes, which are naturally produced by certain bacteria and archaea, cleave double stranded DNA (dsDNA) at specific sequence sites in the DNA. Restriction enzyme (endonuclease) based molecular cloning is the "classic" cloning method, and for many reasons, remains one of the most popular today. The following guide will highlight several of the most popular cloning methods used to create recombinant DNA. Check out our blog on choosing the right cloning method for your research project. This may be due to speed, cost, availability of starting materials or just personal preference. Oftentimes several approaches will work for any specific cloning project however, it is likely that for any given project there is an ideal approach. There are many methods that have been utilized over the years to move around pieces of DNA. With molecular cloning scientists can amplify and manipulate genes of interest and then insert them into plasmids for replication and protein expression. Molecular cloning or the creation of recombinant DNA is an essential process used in scientific research and discovery.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
